|
Replies: 5
| visibility 1
|
Oculus Spirit [98016]
TigerPulse: 100%
Posts: 65066
Joined: 7/13/02
|
|
|
|
 |
CU Medallion [56338]
TigerPulse: 100%
Posts: 31716
Joined: 8/27/02
|
I'm sorry, what are you saying exactly?
Jul 20, 2023, 5:48 PM
|
|
"Nothing new here really, other than Pfizer admitting what everyone knows, some for 3 years now."
What did Pfizer admit that we all knew? The study used the Pfizer/BIONtech vaccine but the authors are from Stanford and other universities. Pfizer isn't saying anything and I didn't know anything about CD8+ cells or CD4+ T cells.
"they just now decided to study T cell responses to the vaccines in people who've had covid?"
The findings were published in March, meaning the study began some time before that. We're only three years into the pandemic, so I'm not sure how quickly you expect things to move. Also, the study isn't about people who have had covid — it's about people who have had covid before being vaccinated.
If you're feeling impatient, buckle up: "That's why, in my mind, it's very important to follow up a study like this, especially when our clinical cohort is getting more years down the road, so that we could be able to correlate that with some more clinical symptoms," Jiang said.
|
|
|
|
|
 |
Oculus Spirit [98016]
TigerPulse: 100%
Posts: 65066
Joined: 7/13/02
|
Instead of typing a dissertation, I will just post some
Jul 20, 2023, 6:40 PM
|
|
links. Feel free to check the dates on these studies.
https://www.frontiersin.org/articles/10.3389/fimmu.2022.1034159/full
https://www.nature.com/articles/s41590-021-01113-x?s=09
https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009742
https://www.frontiersin.org/articles/10.3389/fimmu.2022.843342/full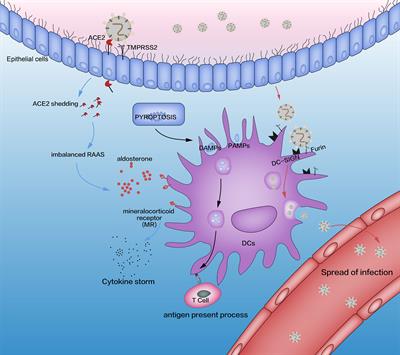 Depletion and Dysfunction of Dendritic Cells: Understanding SARS-CoV-2 Infection Uncontrolled severe acute respiratory syndrome-coronavirus (SARS-CoV)-2 infection is closely related to disorders of the innate immune and delayed adaptive immune systems. Dendritic cells (DCs) “bridge” innate immunity and adaptive immunity. DCs have important roles in defending against SARS-CoV-2 infection. In this review, we summarize the latest research concerning the role of DCs in SARS-CoV-2 infection. We focus on the complex interplay between DCs and SARS-CoV-2: pyroptosis-induced activation; activation of the renin–angiotensin–aldosterone system; and activation of dendritic cell-specific intercellular adhesion molecule 3-grabbing non-integrin. We also discuss the decline in DC number, the impaired antigen-presentation capability, and the reduced production of type-I interferon of DCs in severe SARS-CoV-2 infection. In addition, we discuss the potential mechanisms for pathological activation of DCs to understand the pattern of SARS-CoV-2 infection. Lastly, we provide a brief overview of novel vaccination and immunotherapy strategies based on DC targeting to overcome SARS-CoV-2 infection.
https://www.frontiersin.org/articles/10.3389/fimmu.2021.640644/full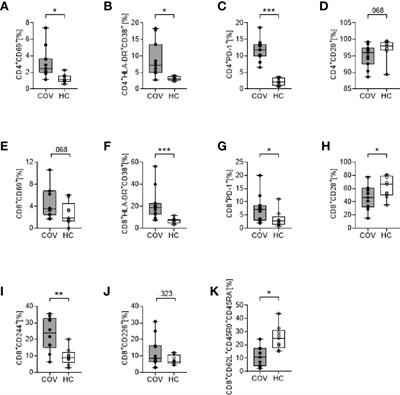 COVID-19 Impairs Immune Response to Candida albicans Infection with SARS-CoV-2 can lead to Coronavirus disease-2019 (COVID-19) and result in severe acute respiratory distress syndrome (ARDS). Recent reports indicate an increased rate of fungal coinfections during COVID-19. With incomplete understanding of the pathogenesis and without any causative therapy available, secondary infections may be detrimental to the prognosis. We monitored 11 COVID-19 patients with ARDS for their immune phenotype, plasma cytokines, and clinical parameters on the day of ICU admission and on day 4 and day 7 of their ICU stay. Whole blood stimulation assays with lipopolysaccharide (LPS), heat-killed Listeria monocytogenes (HKLM), Aspergillus fumigatus, and Candida albicans were used to mimic secondary infections, and changes in immune phenotype and cytokine release were assessed. COVID-19 patients displayed an immune phenotype characterized by increased HLA-DR+CD38+ and PD-1+ CD4+ and CD8+ T cells, and elevated CD8+CD244+ lymphocytes, compared to healthy controls. Monocyte activation markers and cytokines IL-6, IL-8, TNF, IL-10, and sIL2Rα were elevated, corresponding to monocyte activation syndrome, while IL-1β levels were low. LPS, HKLM and Aspergillus fumigatus antigen stimulation provoked an immune response that did not differ between COVID-19 patients and healthy controls, while COVID-19 patients showed an attenuated monocyte CD80 upregulation and abrogated release of IL-6, TNF, IL-1α, and IL-1β toward Candida albicans. This study adds further...
https://www.frontiersin.org/articles/10.3389/fimmu.2022.1080822/full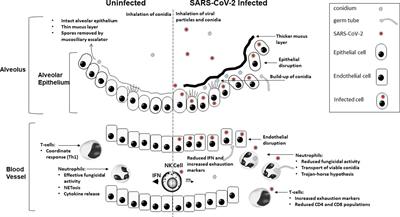 Defective antifungal immunity in patients with COVID-19 The COVID-19 pandemic has placed a huge strain on global healthcare and been a significant cause of increased morbidity and mortality, particularly in at-risk populations. This disease attacks the respiratory systems and causes significant immune dysregulation in affected patients creating a perfect opportunity for the development of invasive fungal disease (IFD). COVID-19 infection can instill a significant, poorly regulated pro-inflammatory response. Clinically induced immunosuppression or pro-inflammatory damage to mucosa facilitate the development of IFD and Aspergillus, Mucorales, and Candida infections have been regularly reported throughout the COVID-19 pandemic. Corticosteroids and immune modulators are used in the treatment of COVID-19. Corticosteroid use is also a risk factor for IFD, but not the only reason for IFD in COVID -19 patients. Specific dysregulation of the immune system through functional exhaustion of Natural killer (NK) cells and T cells has been observed in COVID-19 through the expression of the exhaustion markers NK-G2A and PD-1. Reduced fungicidal activity of neutrophils from COVID-19 patients indicates that immune dysfunction/imbalance are important risk factors for IFD. The COVID-19 pandemic has significantly increased the at-risk population for IFD. Even if the incidence of IFD is relatively low, the size of this new at-risk population will result in a substantial increase in the overall, annual number of IFD cases. It is important to understa...
https://pubmed.ncbi.nlm.nih.gov/32425950/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7779612/
https://www.nature.com/articles/s41392-021-00749-3 SARS-CoV-2 infection causes immunodeficiency in recovered patients by downregulating CD19 expression in B cells via enhancing B-cell metabolism - Signal Transduction and Targeted Therapy The SARS-CoV-2 infection causes severe immune disruption. However, it is unclear if disrupted immune regulation still exists and pertains in recovered COVID-19 patients. In our study, we have characterized the immune phenotype of B cells from 15 recovered COVID-19 patients, and found that healthy controls and recovered patients had similar B-cell populations before and after BCR stimulation, but the frequencies of PBC in patients were significantly increased when compared to healthy controls before stimulation. However, the percentage of unswitched memory B cells was decreased in recovered patients but not changed in healthy controls upon BCR stimulation. Interestingly, we found that CD19 expression was significantly reduced in almost all the B-cell subsets in recovered patients. Moreover, the BCR signaling and early B-cell response were disrupted upon BCR stimulation. Mechanistically, we found that the reduced CD19 expression was caused by the dysregulation of cell metabolism. In conclusion, we found that SARS-CoV-2 infection causes immunodeficiency in recovered patients by downregulating CD19 expression in B cells via enhancing B-cell metabolism, which may provide a new intervention target to cure COVID-19.
NY Times admitted it, then never mentioned it again.
https://www.nytimes.com/2020/06/26/health/coronavirus-immune-system.html
https://www.nature.com/articles/s41392-022-00919-x#citeas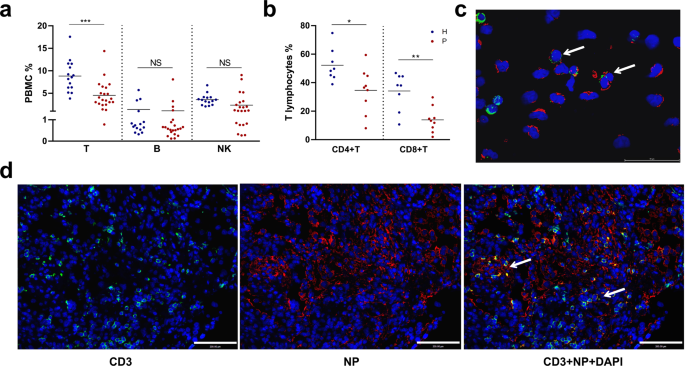 ACE2-independent infection of T lymphocytes by SARS-CoV-2 - Signal Transduction and Targeted Therapy SARS-CoV-2 induced marked lymphopenia in severe patients with COVID-19. However, whether lymphocytes are targets of viral infection is yet to be determined, although SARS-CoV-2 RNA or antigen has been identified in T cells from patients. Here, we confirmed that SARS-CoV-2 viral antigen could be detected in patient peripheral blood cells (PBCs) or postmortem lung T cells, and the infectious virus could also be detected from viral antigen-positive PBCs. We next prove that SARS-CoV-2 infects T lymphocytes, preferably activated CD4 + T cells in vitro. Upon infection, viral RNA, subgenomic RNA, viral protein or viral particle can be detected in the T cells. Furthermore, we show that the infection is spike-ACE2/TMPRSS2-independent through using ACE2 knockdown or receptor blocking experiments. Next, we demonstrate that viral antigen-positive T cells from patient undergone pronounced apoptosis. In vitro infection of T cells induced cell death that is likely in mitochondria ROS-HIF-1a-dependent pathways. Finally, we demonstrated that LFA-1, the protein exclusively expresses in multiple leukocytes, is more likely the entry molecule that mediated SARS-CoV-2 infection in T cells, compared to a list of other known receptors. Collectively, this work confirmed a SARS-CoV-2 infection of T cells, in a spike-ACE2-independent manner, which shed novel insights into the underlying mechanisms of SARS-CoV-2-induced lymphopenia in COVID-19 patients.
https://onlinelibrary.wiley.com/doi/10.1111/all.15372
https://www.biorxiv.org/content/10.1101/2022.05.20.492764v1.full.pdf
https://www.biorxiv.org/content/10.1101/2022.08.18.504053v1.full.pdf
https://www.frontiersin.org/articles/10.3389/fimmu.2021.676932/full Longitudinal Analysis of COVID-19 Patients Shows Age-Associated T Cell Changes Independent of Ongoing Ill-Health ObjectivesThe immunological and inflammatory changes following acute COVID-19 are hugely variable. Persistent clinical symptoms following resolution of initial infection, termed long COVID, are also hugely variable, but association with immunological changes has not been described. We investigate changing immunological parameters in convalescent COVID-19 and interrogate their potential relationships with persistent symptoms.MethodsWe performed paired immunophenotyping at initial SARS-CoV-2 infection and convalescence (n=40, median 68 days) and validated findings in 71 further patients at median 101 days convalescence. Results were compared to 40 pre-pandemic controls. Fatigue and exercise tolerance were assessed as cardinal features of long COVID using the Chalder Fatigue Scale and 6-minute-walk test. The relationships between these clinical outcomes and convalescent immunological results were investigated.ResultsWe identify persistent expansion of intermediate monocytes, effector CD8+, activated CD4+ and CD8+ T cells, and reduced naïurl=https://www.frontiersin.org/articles/10.3389/fimmu.2021.676932/40, median 68 days) and validated findings in 71 further patients at median 101 days convalescence. Results were compared to 40 pre-pandemic controls. Fatigue and exercise tolerance were assessed as cardinal features of long COVID using the Chalder Fatigue Scale and 6-minute-walk test. The relationships between these clinical outcomes and convalescent immunological results were investigated.ResultsWe identify persistent expansion of intermediate monocytes, effector CD8+, activated CD4+ and CD8+ T cells, and reduced naïve CD4+ and CD8+ T cells at 68 days, with activated CD8+ T cells remaining increased at 101 days. Patients >60 years also demonstrate reduced naïve CD4+ and CD8+ T cells and expanded activated CD4+ T cells at 101 days. Ill-health, fatigue, and reduced exercise tolerance were common in this cohort. These symptoms were not associated with immune cell populations or circulating inflammatory cytokines.ConclusionWe demonstrate myeloid recovery but ...
https://www.nature.com/articles/s41418-022-00936-x T cell apoptosis characterizes severe Covid-19 disease - Cell Death & Differentiation Severe SARS-CoV-2 infections are characterized by lymphopenia, but the mechanisms involved are still elusive. Based on our knowledge of HIV pathophysiology, we hypothesized that SARS-CoV-2 infection-mediated lymphopenia could also be related to T cell apoptosis. By comparing intensive care unit (ICU) and non-ICU COVID-19 patients with age-matched healthy donors, we found a strong positive correlation between plasma levels of soluble FasL (sFasL) and T cell surface expression of Fas/CD95 with the propensity of T cells to die and CD4 T cell counts. Plasma levels of sFasL and T cell death are correlated with CXCL10 which is part of the signature of 4 biomarkers of disease severity (ROC, 0.98). We also found that members of the Bcl-2 family had modulated in the T cells of COVID-19 patients. More importantly, we demonstrated that the pan-caspase inhibitor, Q-VD, prevents T cell death by apoptosis and enhances Th1 transcripts. Altogether, our results are compatible with a model in which T-cell apoptosis accounts for T lymphopenia in individuals with severe COVID-19. Therefore, a strategy aimed at blocking caspase activation could be beneficial for preventing immunodeficiency in COVID-19 patients.
https://www.science.org/doi/10.1126/sciimmunol.abe4782
|
|
|
|
|
 |
CU Medallion [56338]
TigerPulse: 100%
Posts: 31716
Joined: 8/27/02
|
Instead of answering basic questions, you will post a
Jul 20, 2023, 10:36 PM
|
|
linkbomb.
You're posting sources for an argument you haven't made. Simply put, what the #### are you talking about?
|
|
|
|
|
 |
Oculus Spirit [98016]
TigerPulse: 100%
Posts: 65066
Joined: 7/13/02
|
Covid damages CD+8 T cells. That's been known for 3 years
Jul 21, 2023, 8:48 AM
|
|
All I was saying. "Nothing new". And yes, it does impact how well you react (the success) of vaccines, of all types. Among other things.
|
|
|
|
|
 |
All-TigerNet [12042]
TigerPulse: 100%
Posts: 10601
Joined: 9/19/01
|
Too bad
Jul 20, 2023, 11:30 PM
[ in reply to Instead of typing a dissertation, I will just post some ] |
|
the US gubmint was complicit with the ChiComs in inflicting this on the world.
Lots of folks need to be in prison or worse. It looks like there will be no accountability.
Ain't that some ####.
|
|
|
|
|
|
Replies: 5
| visibility 1
|
|
|


 to award
the award.
to award
the award.